Nature Knows and Psionic Success
God provides
Developing technology to place minute ‘marks’ in the brain
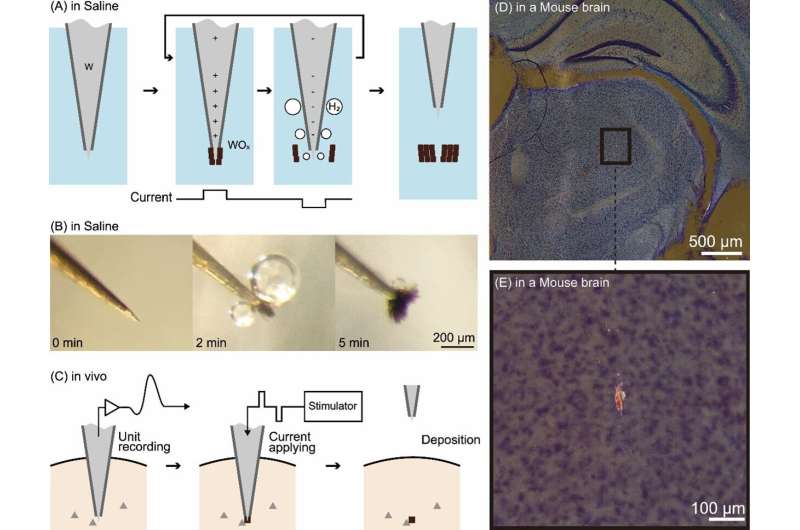
(A) Principle of the technology. (B) Electrolysis of a tungsten electrode in physiological saline. The black lumps are oxides. (C) Procedure for applying this technology in vivo. (D) Dark-field observation image of a slice of a mouse brain in which this technology was applied in deep brain regions. (E) Enlarged view of the black-bounded area in (D). The glowing red mass is an oxide mark. Credit: Toyohashi University of Technology. Researchers have developed a technology capable of deploying very small “marks” in regions of the brain where activity has been recorded.
These marks are created by passing a microcurrent through tungsten electrodes inserted in the brain to record brain activity . These marks do not damage brain tissue and can remain safely inside the brain of a living organism. This technology enables a high-resolution visualization of the brain-wide distribution of neurons with particular functional characteristics. The results of this research were published in eNeuro .
Studies involving recording the activities of deep brain regions, such as the hippocampus and thalamus, commonly utilize needle-like microelectrodes penetrating into the brain to record the electrical activity of neurons. These electrodes have the advantage of higher spatiotemporal resolution than fMRI or EEG recordings and are capable of recording signals from the tissues of deep brain region.
However, one disadvantage of using these electrodes is that it becomes quite difficult to ascertain where exactly in the brain the electrode tip was situated after it has been removed. To address this shortcoming, a mark placement method called “marking” has been widely used. However, these existing methods have limited spatial resolution and cause damage to brain tissue. Therefore, the maximum degree of spatial precision possible with conventional experimental methods remains approximately 0.1 mm.
Tatsuya Oikawa, Toshimitsu Hara, Kento Nomura, along with Associate Professor Kowa Koida of the Department of Computer Science and Engineering, Toyohashi University of Technology, contributed to the study. Oikawa and his colleagues focused on the electrolysis of tungsten, a metal commonly used to make the aforementioned needle-like electrodes. They developed a method wherein electrolysis was employed to deposit oxidized tungsten within the tissues of the deep brain region.
When electrolyzed, the tungsten electrode generates metal oxide and hydrogen bubbles at positive and negative currents, respectively. By rapidly alternating between positive and negative currents, metal oxide is repeatedly generated at the tip of the tungsten electrode, followed by detachment by hydrogen bubbles; this results in the formation of a small lump of oxide. This lump of oxide serves as a mark indicating the precise location of the electrode tip.
Oikawa and his colleagues used the brains of mice and monkeys as models to generate minute oxide lumps (as small as 20 µm in diameter) at the electrode tip and confirmed that they could be deposited within the brain. They also confirmed that the current used to generate these marks and the deposited oxides do not damage brain tissue.
Furthermore, they made the noteworthy discovery that the deposited oxides appear to glow red when stained using a standard brain tissue stain (Nissl staining) and observed under a microscope using dark-field illumination. This distinctive characteristic clearly differentiates the marks from surrounding noise, facilitating the easy identification of small markings through low-power microscopy. Development background
Associate Professor Kowa Koida, the advisor to Oikawa and his colleagues, said, “Originally, our plan was to remove plated coatings applied in advance to the electrodes and utilize them as marks. Unfortunately, a mix-up occurred during experimentation, leading us to unintentionally employ an incompletely plated electrode. Surprisingly, despite this setback, we observed the formation of a mark. Upon closer consideration, we recognized that electrolysis, which is commonly used to form tungsten rods into thin needles for electrodes, produces an oxide powder.”
“That powder can become a mark. We also realized that electrolysis can be performed in vivo. Furthermore, we found that the current parameters necessary for electrolysis can align with those employed when activating neurons through current delivered via the electrode. Essentially, the safety of the organism marked using our method has already been validated in previous experiments involving current stimulation. While it has been widely known that current stimulation results in the wear and tear of electrode tips, what remained unnoticed was that simultaneously, metal oxides are deposited in the body, serving as marks.” Future outlook
Now, Oikawa and his colleagues are applying this technology to unravel the microscopic functional structure of the lateral geniculate nucleus, a visual center deep in the brain of macaque monkeys. By providing examples of effective applications of this technology, they aim to foster its widespread adoption as a fundamental technique in neuroscience.
Furthermore, this processing technique, utilizing in vivo metal electrolysis for deposition, has been used in the medical field, such as the treatment of aneurysms using embolization coils. Therefore, electrolysis in vivo has already garnered a certain level of trust. By combining both the measurement and deposition processes into a unified technique using microelectrodes, the technologies used in this study may be applied to medical care as well.
Provided by Toyohashi University of Technology
Read more at medicalxpress.com
