Nature Knows and Psionic Success
God provides
Huntington’s spreads like ‘fire in the brain.’ Scientists say they’ve found the spark
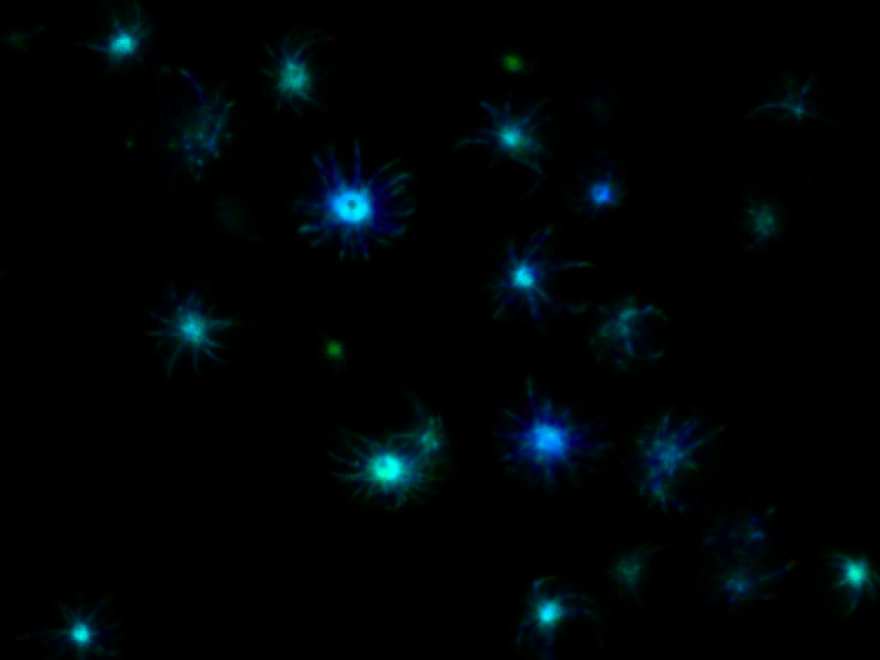
In Huntington’s disease, proteins form toxic clumps that kill brain cells. Diseases like Alzheimer’s, Parkinson’s, and Huntington’s are caused by toxic clumps of proteins that spread through the brain like a forest fire.
Now scientists say they’ve figured out how the fire starts in at least one of these diseases. They’ve also shown how it can be extinguished.
The finding involves Huntington’s disease, a rare, inherited brain disorder that cut short the life of songwriter Woody Guthrie . But the study has implications for other degenerative brain diseases, including Alzheimer’s.
It “opens the path” to finding the initial event that leads to diseases like Alzheimer’s and Parkinson’s, says Corinne Lasmézas , who studies neurodegenerative diseases at the Wertheim UF Scripps Institute in Jupiter, Florida. She was not involved in the study.
People with Huntington’s “begin to lose control of their body movements, they have mental impediments over time, and eventually they die,” says Randal Halfmann , an author of the study and a researcher at the Stowers Institute for Medical Research in Kansas City, Mo.
Like other neurodegenerative diseases, Huntington’s occurs when proteins in the brain fold into an abnormal shape and begin to stick together. Then these clumps of abnormal protein begin to cause nearby proteins to misfold and clump too.
“As the disease progresses you’re effectively watching a sort of a forest fire,” Halfmann says. “And you’re trying to figure out what started it.”
In essence, Halfmann’s team wanted to find the molecular matchstick responsible for the lethal blaze. Looking inside a cell
To do that, they needed to chronicle an event that is fleeting and usually invisible. It’s called nucleation , the moment when a misfolded protein begins to aggregate and proliferate.
The team developed a way to conduct experiments inside individual cells. They used genetic tweaks to create hundreds of versions of a protein segment called PolyQ, which becomes toxic in Huntington’s.
The team placed different versions of PolyQ in a cell, then look for signs of misfolding and clumping.
“It’s sort of like if you’re in a dark room and you’re trying to figure out what the shape of the room is,” Halfmann says. “You just keep bumping into things and eventually you bump into things enough times to figure out exactly what it looks like.”
The trial-and-error approach worked, Halfmann says. “What starts this little forest fire in the brain is a single molecule of PolyQ.”
Once the team had identified that molecule, they were able to find a way to prevent it from spreading — at least in the lab. The trick was to flood the cell with proteins that, in effect, smothered the flame before it could do any damage.
The next step will be to develop a drug that can do something similar in people, Halfmann says.
“Ultimately, it only matters if we actually create a therapy,” he says. “Otherwise, it’s just academics.”
The study could also lead to new treatments for other neurodegenerative diseases, Lasmézas says, treatments that prevent the cascade of events that leads to brain damage.
“You have to go back when the fire starts, so that it doesn’t propagate in the entire forest,” she says. Lessons for Alzheimer’s research?
The Alzheimer’s field appears to be learning that lesson.
Early drugs targeted the large amyloid plaques found in the brains of people with the disease. But these drugs didn’t work , perhaps because the plaques they sought to eliminate are just the charred remains of a forest that has already burned.
Lasmézas says the latest drugs, like lecanemab , still remove large clumps of amyloid, “but they also recognize the ones that are smaller and that are more toxic. And this is why they block more efficiently, the neuronal toxicity.”
These smaller clumps form before plaques appear, and are closer to the event that touches off Alzheimer’s in the first place, Lasmézas says.
Studies like the one on Huntington’s show that scientists are finally closing in on strategies that will slow or halt diseases including Parkinson’s and Alzheimer’s, Lasmézas says.
“For a long time, we didn’t know much about the mechanism of neurodegenerative diseases,” she says. “Within the last, let’s say, 15 years, there’s been literally an explosion of knowledge.”
Copyright 2023 NPR. To see more, visit https://www.npr.org.
NPR Top News
